This procedure covers the basic concepts for high-quality soldering.
Procedure
Soldering Process
Soldering is the process of joining two metals using a solder alloy, and it is one of the oldest known joining techniques. Faulty solder joints remain one of the major causes of equipment failure; thus, the importance of high standards of workmanship in soldering cannot be overemphasized. The guide covers basic soldering procedures and has been designed to provide the fundamental knowledge needed to complete most high-reliability hand soldering and component removal operations.
Properties of Solder
Solder, used for electronics, is a metal alloy that combines various elements in different proportions. These proportions are usually marked on the various types of solder available. With many solders, melting does not occur simultaneously. For example, 50/50 tin/lead solder begins to melt at 183 C (361 F) but is not fully melted until the temperature reaches 216 C (420 F). Between these two temperatures, the solder exists in a plastic or semi-liquid state. The plastic range of a solder varies depending upon the ratio of elements. With 60/40 tin/lead solder, the range is much smaller than 50/50 solder. The 63/37 ratio, known as eutectic solder, has practically no plastic range and melts almost instantly at 183 C (361 F). Movement during the solidification may cause what is known as a disturbed joint. A disturbed joint has a rough, irregular appearance and looks dull instead of bright and shiny. A disturbed solder joint may be unreliable and may require rework.
The following table lists common solders business for electronics soldering applications.
| Solder Material Type |
Melting Temperature |
| Tin-Lead (Sn-Pb) |
183-188°C (361-370°F) |
| Lead-Free (Sn-Ag-Cu) |
217-220°C (422-428°F) |
| High-Temperature (Sn-Ag) |
221°C- Up (430°F - Up) |
Wetting Action
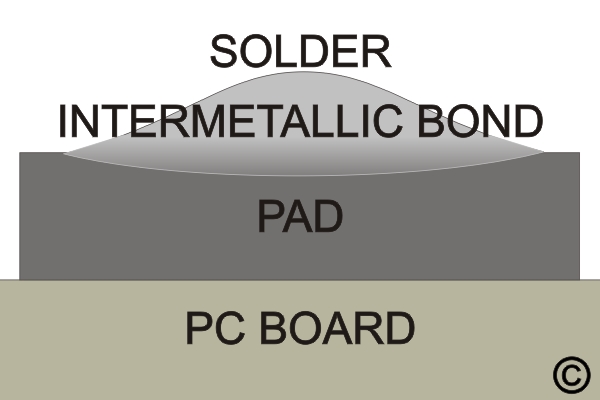
A metal-solvent action occurs when the hot solder comes in contact with a copper surface. The solder dissolves and penetrates the copper surface. The molecules of solder and copper blend to form a new alloy, one that's part copper and part solder. This solvent action is called wetting, forming the intermetallic bond between the parts. (See Figure 1) Wetting can only occur if the surface of the copper is free of contamination and from the oxide film that forms when the metal is exposed to air. Also, the solder and work surface must have reached the proper temperature. Although the surfaces to be soldered may look clean, there is always a thin film of oxide covering it. For a good solder bond, surface oxides must be removed during the soldering process using flux.
Flux
Reliable solder connections can only be accomplished with truly cleaned surfaces. Solvents can be used to clean the surfaces before soldering but are insufficient due to the extremely rapid rate at which oxides form on the surface of heated metals. To overcome this oxide film, using materials called fluxes in electronic soldering becomes necessary. Fluxes consist of natural or synthetic rosins and sometimes chemical additives called activators.
The flux's function is to remove oxides and keep them removed during the soldering operation. This is accomplished by the flux action, which is very corrosive at solder melt temperatures and accounts for flux's ability to remove metal oxides rapidly. In its unheated state, however, rosin flux is non-corrosive and non-conductive and thus will not affect the circuitry. The fluxing action of removing oxides, carrying them away, and preventing the reformation of new oxides allows the solder to form the desired intermetallic bond.
Flux must melt at a temperature lower than solder to do its job before the soldering action. It will volatilize rapidly; thus, it is mandatory that flux be melted to flow onto the work surface and not simply volatilized by the hot iron tip to provide the full benefit of the fluxing action. There are varieties of fluxes available for many purposes and applications. The most common types include Rosin - No Clean, Mildly Activated, and Water Soluble.
When used, the liquid flux should be applied in a thin, even coat to those surfaces being joined before applying heat. Cored wire solder and solder paste should be placed so that the flux can flow and cover the joints as the solder melts. Flux should be applied to prevent damage to the surrounding parts and materials.
Soldering Irons
Soldering irons come in a variety of sizes and shapes. A continuously tinned surface must be maintained on the soldering iron tip's working surface to ensure proper heat transfer and avoid impurities transfer to the solder connection.
Before using the soldering iron, the tip should be cleaned by wiping it on a wet sponge. When not in use, the iron should be kept in a holder, with its tip clean and coated with a small amount of solder. Although tip temperature is not the key element in soldering, you should always start at the lowest temperature possible. A good rule of thumb is to set the soldering iron tip temperature at 260 C - 500 F and increase the temperature as needed to obtain the desired result.
Controlling Heat
Controlling soldering iron tip temperature is not the key element in soldering. The key element is controlling the heat cycle of the work. How fast the work gets hot, how hot it gets, and how long it stays hot are the elements to control for reliable solder connections.
Thermal Mass
The first factor that needs to be considered when soldering is the relative thermal mass of the joint to be soldered. This mass may vary over a wide range. Each joint has a particular thermal mass, and how this combined mass compares with the iron tip's mass determines the work's time and temperature rise.
Surface Condition
The second factor of importance when soldering is the surface condition. If any oxides or other contaminants cover the pads or leads, there will be a barrier to the heat flow. Even though the iron tip is the right size and temperature, it may not be able to supply enough heat to the joint to melt the solder.
Thermal Linkage
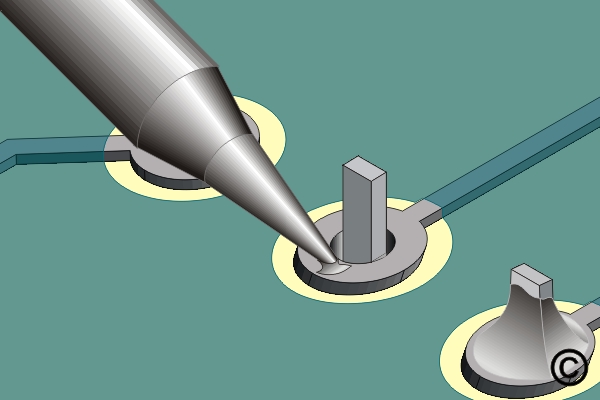
A third factor to consider is thermal linkage. This is the area of contact between the iron tip and the work. Figure 2 shows a view of a soldering iron tip soldering a component lead. Heat is transferred through the small contact area between the soldering iron tip and pad. The thermal linkage area is small.
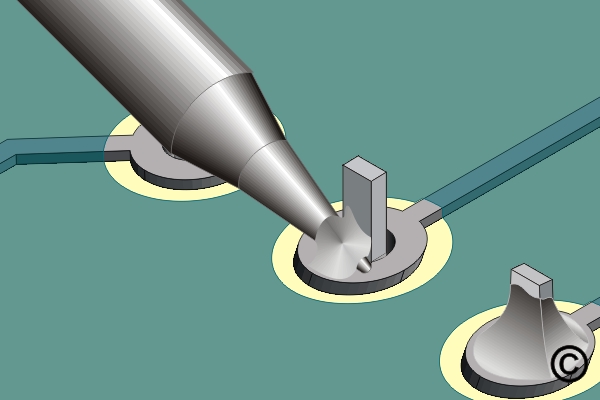
Figure 3 also shows a view of a soldering iron tip soldering a component lead. In this case, the contact area is greatly increased by having a small amount of solder at the point of contact. The tip is also in contact with both the pad and component, further improving the thermal linkage. This solder bridge provides thermal linkage and assures the rapid transfer of heat into the work.
Applying Solder
Generally, the soldering iron tip should be applied to the maximum mass point of the joint. This will permit the rapid thermal elevation of the parts to be soldered. Molten solder always flows from the cooler area toward the hotter one.
Before solder is applied, the surface temperature of the soldered parts must be elevated above the solder melting point. Never melt the solder against the iron tip and allow it to flow onto a surface cooler than the solder melting temperature. Solder applied to a cleaned, fluxed, and properly heated surface will melt and flow without direct contact with the heat source and provide a smooth, even surface, filleting out to a thin edge. Improper soldering will exhibit a built-up, irregular appearance and poor filleting. For good solder joint strength, soldered parts must be held in place until the solder solidifies.
If possible, apply the solder to the upper portion of the joint so that the work surfaces, not the iron, will melt the solder and so that gravity will aid the solder flow. Selecting cored solder of the proper diameter will help control the amount of solder being applied to the joint. Use a small gauge for a small joint and a large gauge for a large joint.
Post Solder Cleaning
When cleaning is required, flux residue should be removed immediately but no later than one hour after soldering. Some fluxes may require more immediate action to facilitate adequate removal. The cleaning solution may use mechanical means such as agitation, spraying, brushing, and other application methods. The cleaning solvents, solutions, and methods used should not have affected the parts, connections, and materials being cleaned. After cleaning, boards should be adequately dried.
Resoldering
Care should be taken to avoid the need for resoldering. When resoldering is required, quality standards for the resoldered connection should be the same as for the original connection. A cold or disturbed solder joint will usually require only reheating and reflowing of the solder with the addition of suitable flux. If reheating does not correct the condition, the solder should be removed and the joint resoldered.
Workmanship
Solder joints should have a smooth appearance. The joints should be free from scratches, sharp edges, grittiness, looseness, blistering, or other evidence of poor workmanship. Probe marks from test pins are acceptable, as they do not affect the integrity of the solder joint. An acceptable solder connection should indicate evidence of wetting and adherence when the solder blends to the soldered surface. The solder should form a small contact angle, indicating a metallurgical bond and metallic continuity from solder to surface. (See Figure 4) Smooth, clean voids or unevenness on the surface of the solder fillet or coating are acceptable. A smooth transition from pad to component lead should be evident.